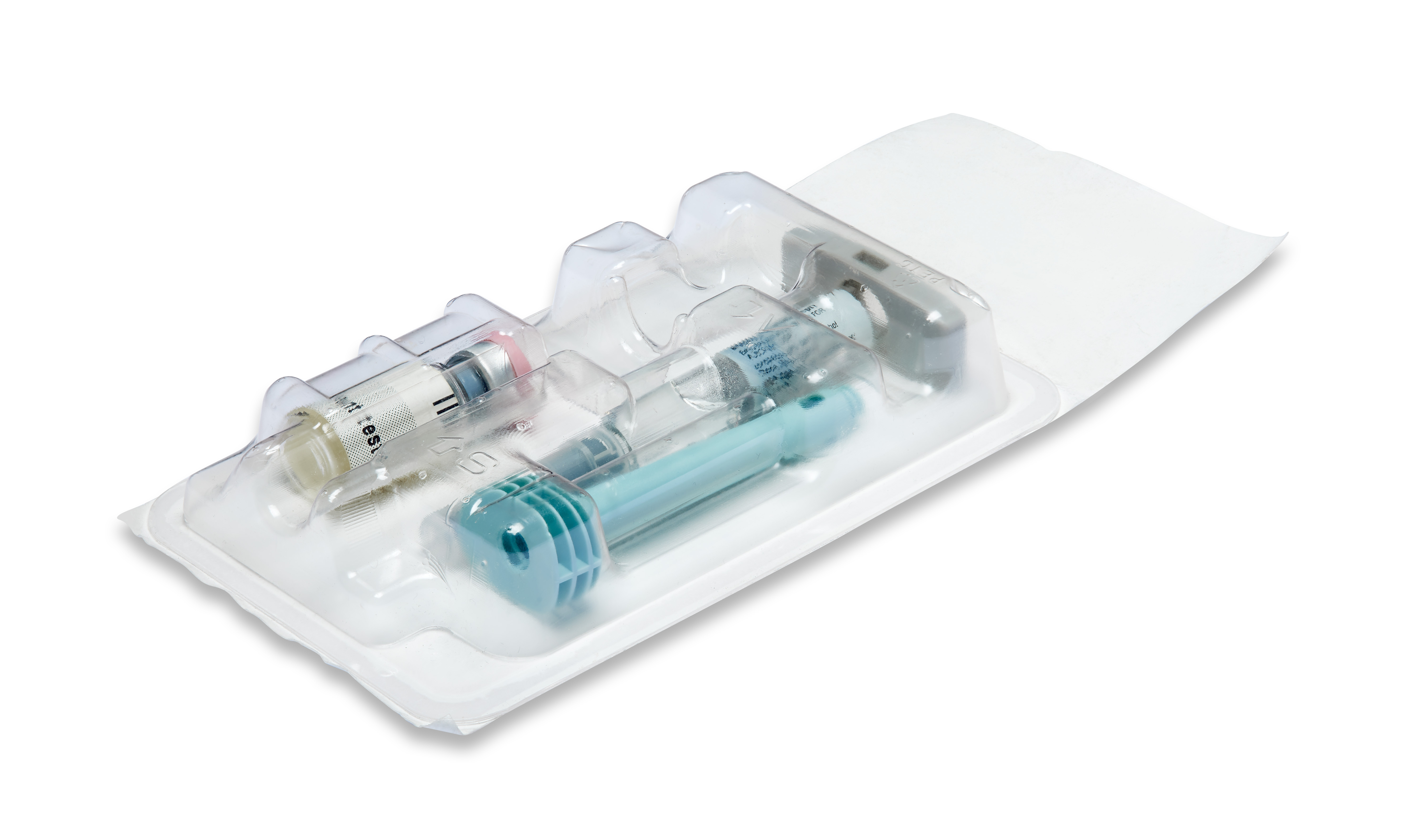
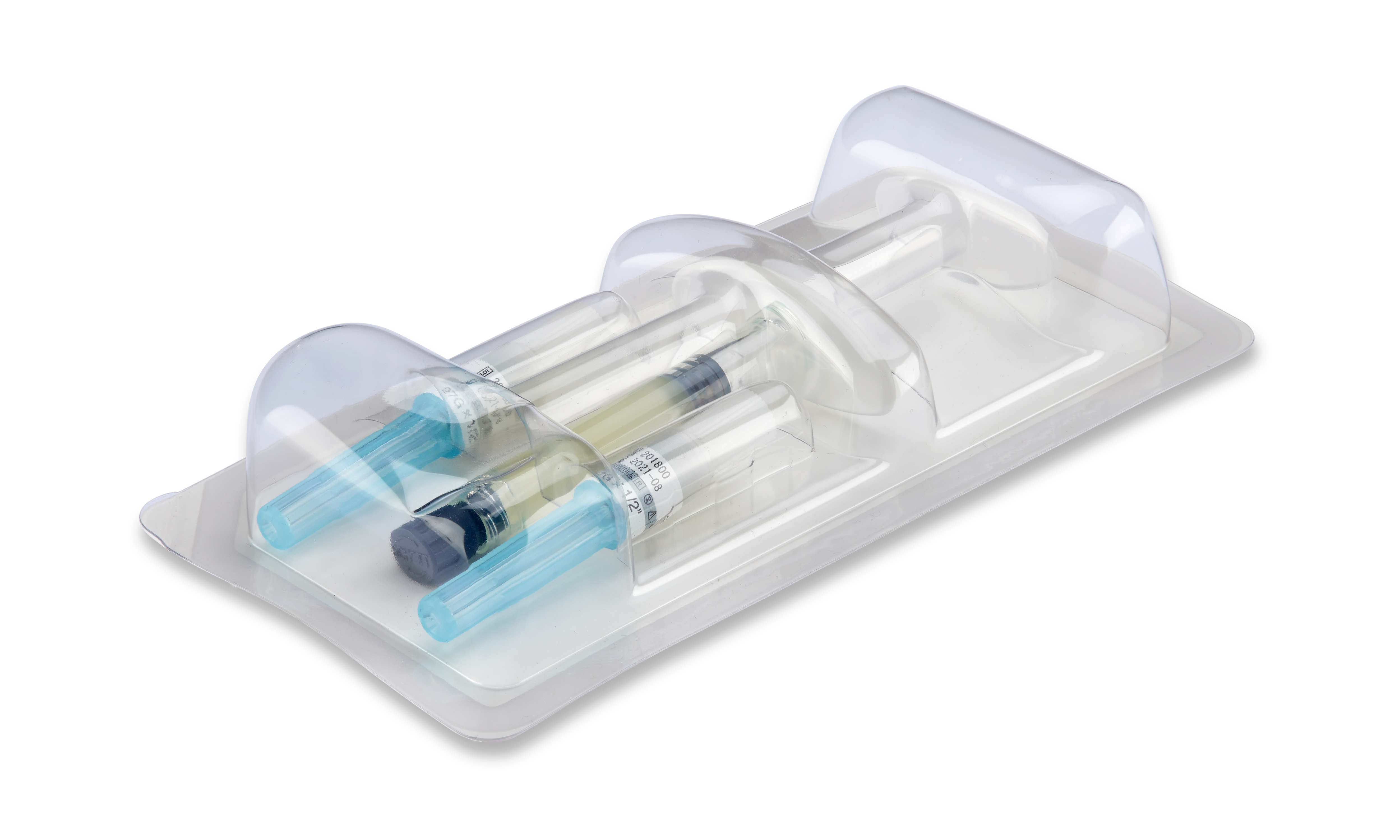
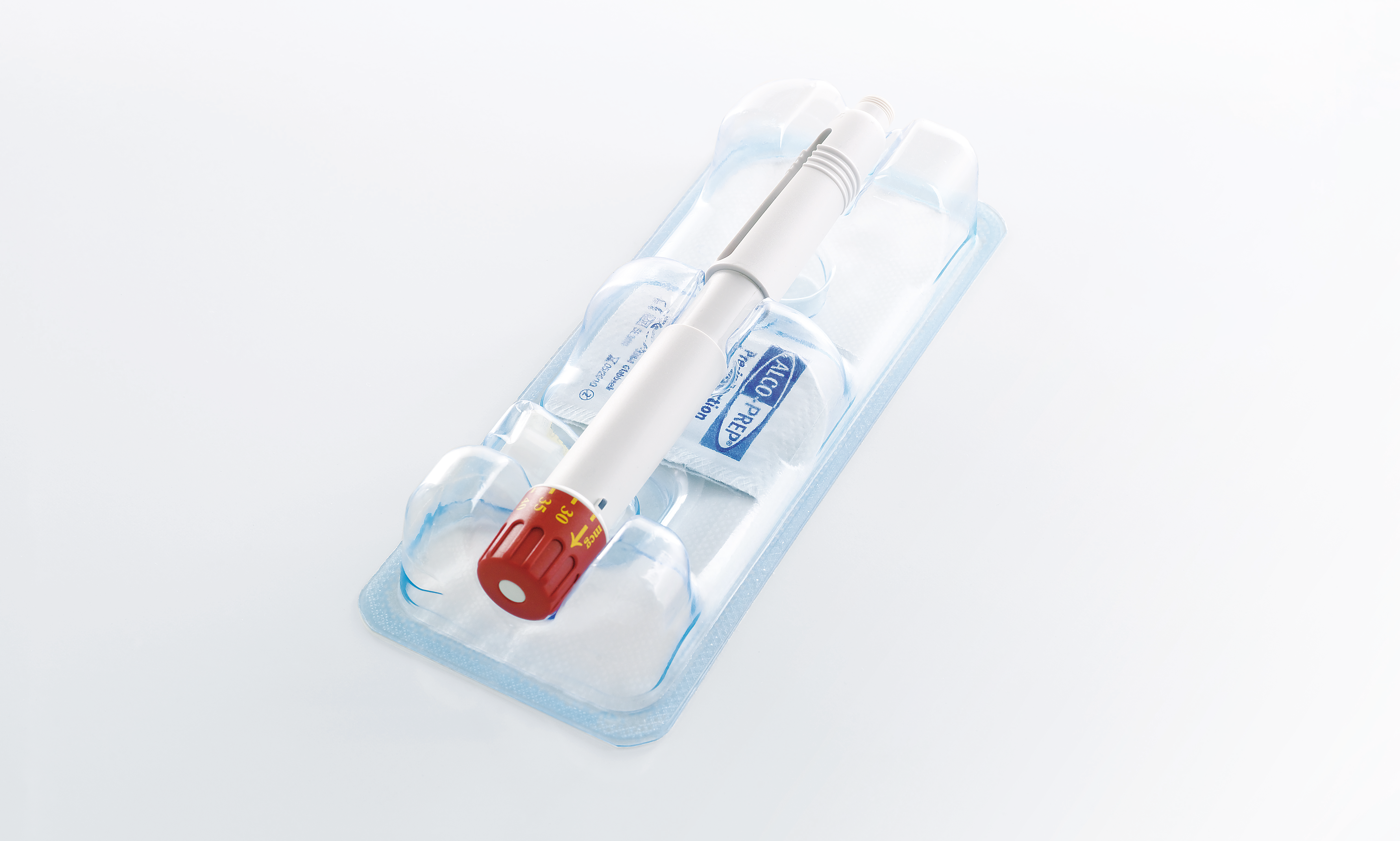
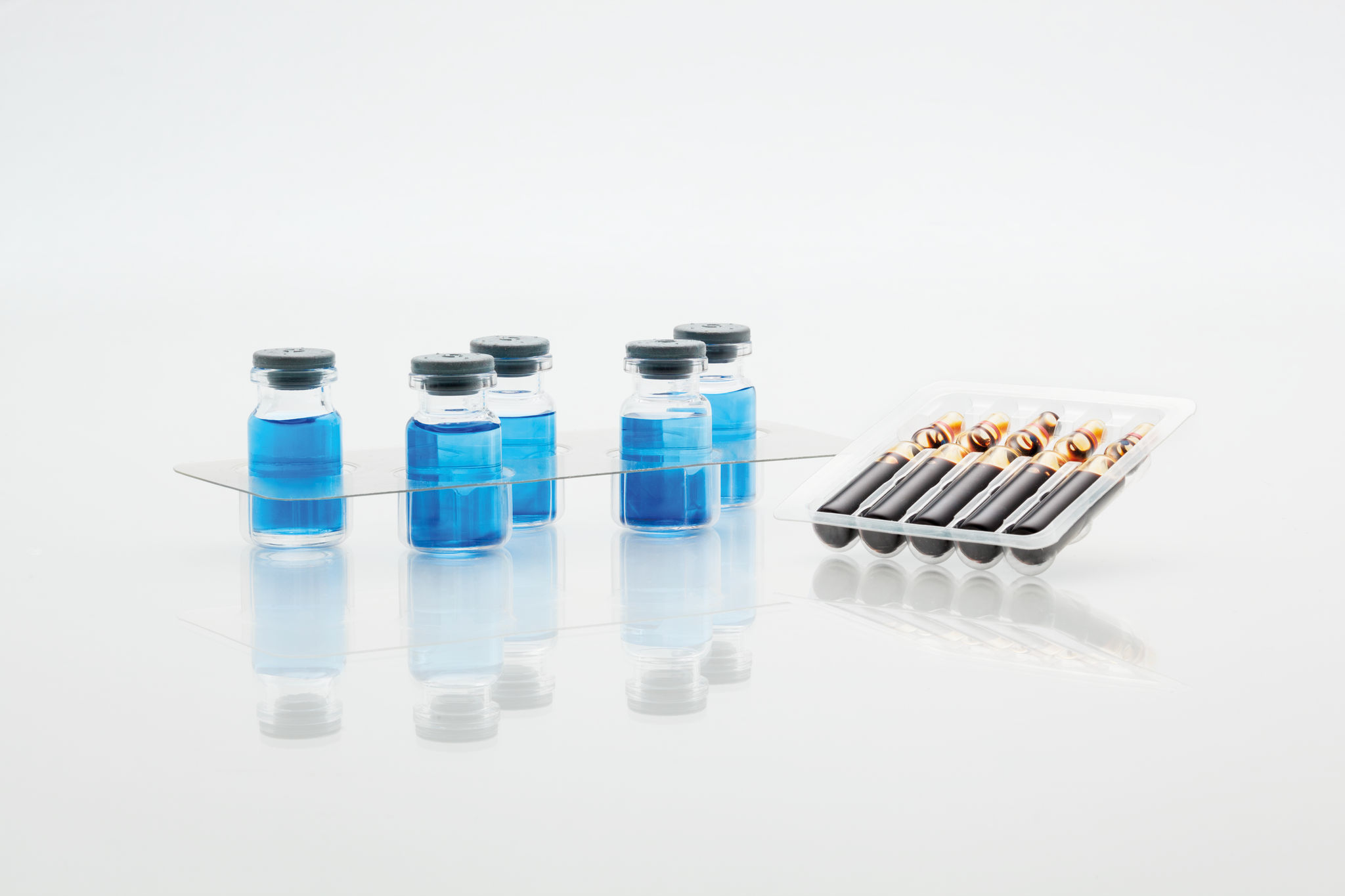
Maximum product and process reliability along the entire line
Process reliability, traceability and product safety are the key requirements in the pharmaceutical industry. Fully automated, individually integrated packaging lines have to meet the most stringent standards for packing syringes, vials, ampoules, auto-injectors, active stents, pens and many other pharmaceutical products. All machines integrated into our line solutions for pharmaceutical products feature GMP-compliant design. Thanks to the seamless linking of all line components, our line solutions offer optimum operating and process reliability as well as maximum efficiency. We can cover the entire value chain, including up- and downstream equipment. On request, we can also integrate third-party products from proven manufacturers with the same outstanding quality requirements as MULTIVAC - and we can even supply you with turnkey solutions.
Many years of proven line expertise ensure process reliability and product safety
Reproducible and traceable processes, correct labelling and marking, cleanroom compatibility and conformity with current guidelines are some of the challenges facing packaging lines for pharmaceutical products. In-depth consulting and joint development are always at the forefront so that we can configure and customize the best possible solution for you. We integrate all modules and technologies into complete solutions based on our comprehensive expertise in packaging lines. We focus on your strategic objectives, safety requirements and individual processes and process sequences. A turnkey solution can be offered on request.
Success stories
 MULTIVAC Packaging Machine for Medical Supplies
MULTIVAC Packaging Machine for Medical Supplies
 MULTIVAC Packaging Machine in Action
MULTIVAC Packaging Machine in Action